Nervous system & thermography: how to measure the training adaptation
In a recent study, Sillero et al. (2022) wanted to investigate the relationship between the nervous system and the temperature of the skin through thermography and the Omegawave system, after a strength training session. They found a significant relationship between a higher temperature rise in those with a less active sympathetic system before the test, which could be used for an indirect assessment of the nervous system.
The relationship between skin temperature and the nervous system is a proven fact in the physiological regulation of body systems. From the hypothalamus-pituitary-thyroid axis (mainly from the main integrator of thermoregulation, the preoptic area of the hypothalamus) the thermal responses to external agents are controlled and detected by the thermoreceptors: Ruffini corpuscles, which detect heat, and Krause corpuscles, which detect the cold.
When the thermal balance is broken, the brain, through the sympathetic nervous system, sets in motion a series of mechanisms to recover the body to its state of homeostasis. For example, before an increase in body temperature there is a vasodilation produced by the cholinergic system that increases peripheral blood flow and allows heat to be dissipated through perspiration.
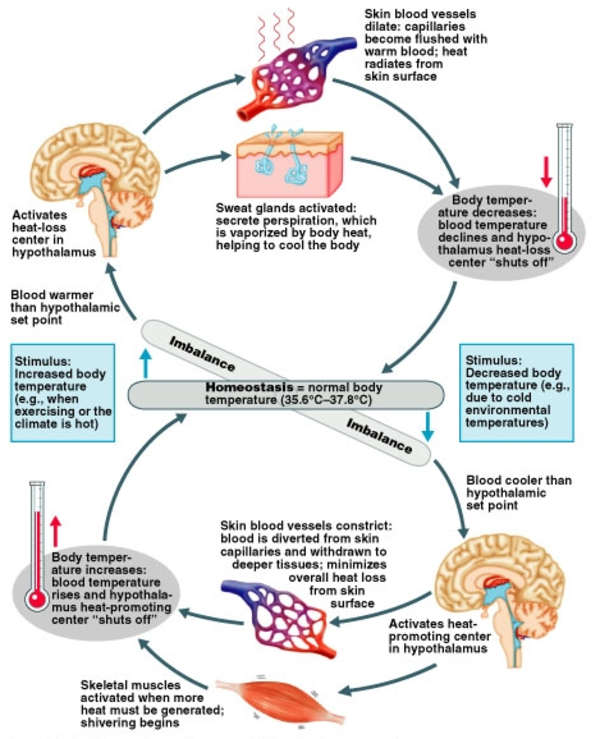
Reflex thermoregulation produces two major phenomena: the thermoregulatory response to cold and the thermoregulatory response to heat:
- The thermoregulatory response to cold is an integrated combination of automatic responses including sympathetic thermogenesis in brown adipose tissue, shivering thermogenesis in skeletal muscle, increased production of expired carbon dioxide by increased metabolic activity, and, lastly, tachycardia.
- The thermoregulatory response to heat on its part includes cutaneous vasodilation, tachypnea, and sweating.
The nervous system has various strategies to recover from an event that triggers an external thermoregulatory response. But internally, the strategies that the body chooses to resolve an increase or decrease in temperature due to an endogenous agent, such as exercise, are still unknown.
Temperature and central nervous system after strength training
In a recent investigation by the research group of Dr. Sillero et al. (2022), they wanted to analyze how strength training affects the response of skin temperature measured with thermography and the activation of the autonomic nervous system by means of the Omegawave device.
To do this, the researchers performed a pre-training evaluation where they analyzed the perceived exertion at rest using the RPE scale, skin temperature with two thermographic images for the anterior and posterior protocol following the TISEM consensus, and the activation of the autonomic nervous system by through the different variables provided by the Omegawave system.
Afterwards, the subjects performed the training that consisted of three exercises: squat on the Panatta “power squat” machine, knee extension on the machine and knee flexion on the machine, all exercises were performed at 70% of the maximum repetition, to complete a total of 4 sets of 10 repetitions. Immediately afterwards, the variables of the first evaluation were recorded again.
After 20 minutes of recovery, the last evaluation of the variables described above was carried out. The perceived exertion (RPE) immediately after training marked a value of 6.75 ± 1.18 on average for all participants and the training time was 30 minutes in total.

The results of the study showed a significant relationship.
Skin temperature showed a significant increase in those areas that were exercised the most. The authors pointed out that the values of skin temperature before exercise can be a predictor of the total increase that the exercised area will suffer with a correspondence in the mathematical model of r2= 63.7% for the anterior thigh and of r2 = 64.4% for the posterior thigh. As seen in previous blog posts, the temperature for analytical exercises increases in the exercised area as a consequence, among other things, of the dissipation of energy in the form of heat and increased fatigue.
In the same way, for the activation of the autonomous system, the variables that were taken into account were those derived from the direct current potential, which established a scale from 0 to 1 (the closer to 0, the less activation and the closer to 1, the greater activation) for the sympathetic and parasympathetic systems.
Sympathetic system values have a highly significant inverse relationship with parasympathetic system values, as expected. Therefore, the authors divided the participants
It is in two groups to look for the influence of the nervous system in the variation of skin temperature after exercise. The groups were established according to whether they were above or below the level of sympathetic activation in the initial analysis (the cut-off point was established at 0.53, which corresponded to the median sympathetic activation). In this way, subjects were divided into those with low activation (≤0.53; n = 10) and those with high activation (>0.54; n = 10).
The authors found that, regardless of the variability of the interpersonal response to exercise, the group with a less active sympathetic system increased the temperature of the exercised regions more (+0.29 ºC) after training than the one with a greater activation of the sympathetic nervous system.

This could show that the subjects with lower activation could be in a different vascular condition before starting the exercise and that the effects of activation would be greater for this group.
Investigations by Kingsley et al (2008) demonstrated that post-exercise muscle ischemia increases vascular sympathetic modulation through baroreflex activation induced by local metabolites in exercising muscle, and elevated blood pressure reactivates baroreflex sensitivity and modulation. cardiac parasympathetic during this condition. This, related to the research by Sillero et al. (2022), shows us that 20 minutes are not enough to reach pre-exercise levels and be fully recovered.
As was the case with temperature, the activation values of the sympathetic nervous system are an independent predictive indicator of the total increase in temperature in the exercised areas.
Conclusions
Skin temperature measured with thermography could be used to estimate the level of participation of muscle groups during exercise. With the consideration that the time between the end of the exercise and the taking of the image has to be taken into account to interpret the data accurately.
The initial activation level of the sympathetic system could influence the response to resistance exercise. How the athlete is prior to exercise (if they have recovered and/or different levels of activation) could lead to different thermal responses of the skin. Recording the level of sympathetic activation before exercise could be interesting to predict the athlete’s response to strength training. Lower activation values predispose to have a higher temperature response and better adaptation.
In addition, the level of sympathetic activation inversely affects the thermal response of the skin, being a significant predictor of the total variation in skin temperature only in the exercised muscle areas.

As a reflection of the study from the ThermoHuman R+D+i department, we understand that athletes who previously come with a lower activation of the sympathetic system (understanding that they are in an advantageous situation because they are more recovered and prepared for the activity) will have a greater thermal response to strength training and, therefore, a greater adaptation to training and a better predisposition to tolerate the training load.
REFERENCES
Sillero-Quintana M, Jones-Rando J, Refoyo I, Marins JCB, Seixas A. Effects of Resistance Training on Skin Temperature and Its Relationship with Central Nervous System (CNS) Activation. Healthcare (Basel). 2022 Jan 21;10(2):207. doi: 10.3390/healthcare10020207. PMID: 35206822; PMCID: PMC8871941.
Kingsley, J.D.; Figueroa, A. Acute and training effects of resistance exercise on heart rate variability. Clin. Physiol. Funct. Imaging
2016, 36, 179–187.